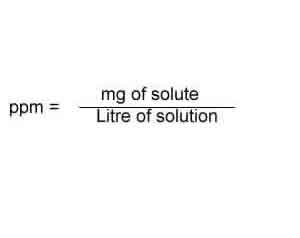
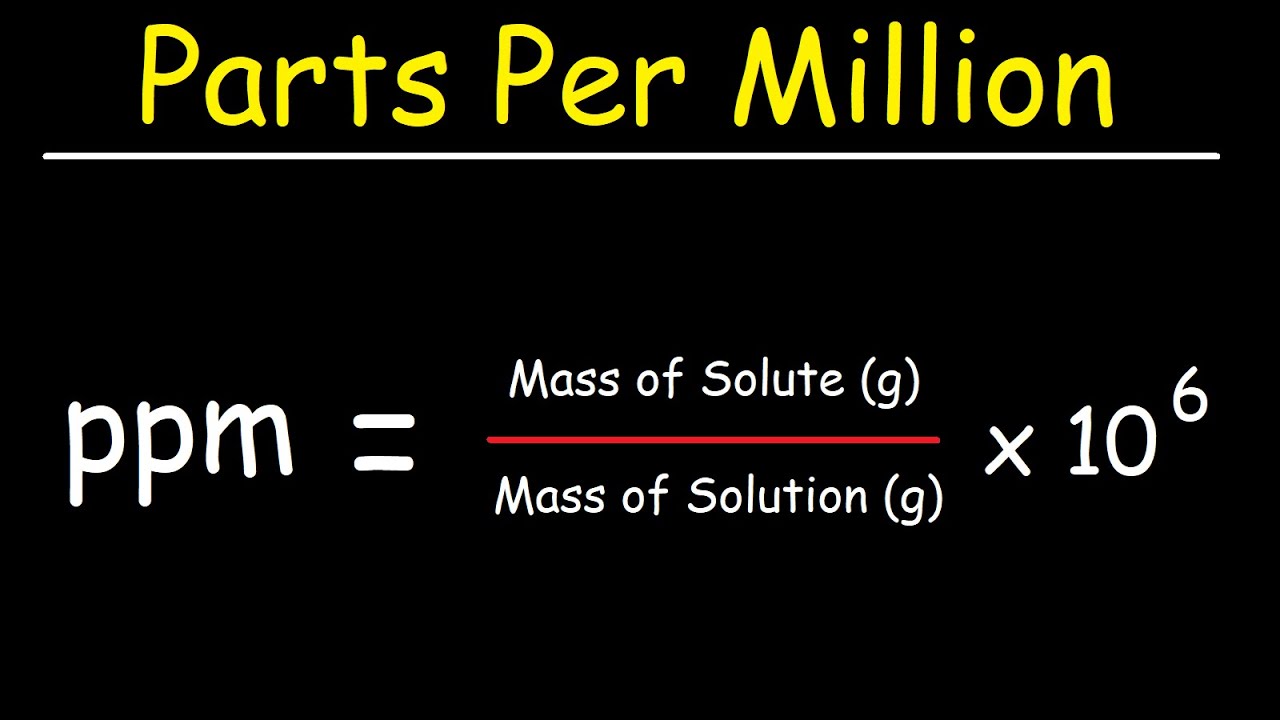A 1 gram of salt in 1 m3 of solution hasconcentration of 1 ppm.ppm is defined as number of parts by mass ofsolute per million parts of solution.

Molarity, Molality, Normality, Part per million (ppm) and other basic terms of Concentration solution with definition & formula |Chemistry Basic|02 | by Amrita Shetty | Medium